BORDERLINE PRODUCTS: MEDICAL DEVICES / MEDICINAL PRODUCTS as per MDR
Thanks to new recommendations provided by the European Commission’s Medical Device Coordination Group , developers trying to determine whether their items are medicines or devices now have better definitions (MDCG).It is quite simple to determine if a product is a device covered by the Medical Devices Regulation 2017/745 or a medication covered by Directive 2001/83/EC on the Community law related to medicinal items for human use (MPD).
Other items, such as medical devices that include a substance that would otherwise be designated a drug, aren’t always apparent whether they’re covered by the MDR or the MPD.By offering precise definitions of various types of products and clarifying how they differ, the newMDCG指南document aims to clarify how developers may find the suitable regulatory framework for these so-called “borderline” scenarios.
Definition:
边界产品是尚不清楚它们从一开始就属于MDR还是MDP的产品。If the product is subject to MDR, it must comply with MD Article 2(1) and must not be within the scope of MDR Article 1(6).
- The team of Operon strategist guide their clients to classify their product and provide regulatory services according to it . AS aCE mark medical devices consultant,我们正在为全球客户提供优质的服务。如果您想利用我们的任何服务联系我们或者whatsapp usyour requirement, will be happy to serve you.
医疗设备附件第2条第2款MDR
为了使医疗设备被视为配件,必须按照其原始目的或明确的目的使用,并直接根据所需的用途来增强医疗设备的医疗功能。
药品(第1(2)条MDR)
Any chemical or combination of substances that is claimed to have the ability to treat or prevent illness in humans.
如何根据MDR确定产品是否满足医疗设备的定义?
Flowchart for determining if a product fulfills the definition of a medical device.
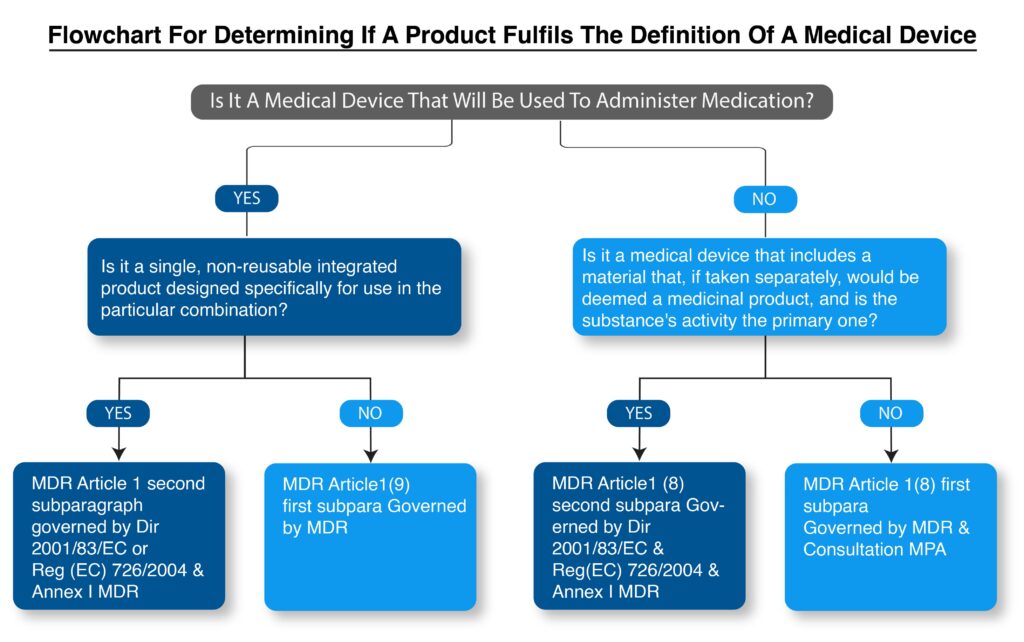
Integral combinations of medical devices and medicinal products are governed as medicinal products.
Under this category contains devices designed to administer a medicinal product and in which the device and the combination of drugs are combined into a single integral component that is intended only for use in the specified combination and is not reusable (MDR, Article 1(9) second subparagraph).它还包括将药物作为整体元素的设备,当药物项目的动作比设备的动作更为重要时(MDR,第1(8)条第二语句)。
作为药物产品调节的整体医疗设备和药品组合就是一些例子。
– Aerosols containing a medicinal product
- 预先装有特定药物的雾化器
– Patches for transdermal drug delivery
– Syringes prefilled with a medicinal product
用于管理药品产品的医疗设备
Specially this category applies to devices that are designed to dispense a medical product within the meaning of the MPD but are not integral to the medicinal product.
在这种情况下,尽管有指令2001/83/EC关于医疗产品的规则,MDR适用于该设备。
Examples of medical devices for administration of medicinal products
– Drug delivery pumps
- 植入式输液泵
– Reusable iontophoresis devices.
将医疗设备纳入必不可少的组成部分,是一种辅助药物
MDR定义了医疗设备的特定实例,该实例包括作为不可或缺的一部分物质,该物质将在MPD的第1条下单独使用,包括MPD第1条的药用产品,包括源自人血或人血浆的药物。, with a secondary action to the device’s primary action.
为了简单起见,本文将其称为包括辅助医学产品作为整体元素的设备。还包括草药。
Examples of medical devices incorporating, as an integral part, an ancillary medicinal product
- Catheters coated with heparin or an antibiotic agent
- Bone cements containing antibiotic
- Root canal fillers which incorporate medicinal products with ancillary action to that of the device
- 软组织填充剂,结合了局部麻醉药
- Bone void filler containing growth factors
- Condoms coated with spermicides
- 带类固醇尖端的电极
- 伤口敷料,手术或屏障窗帘(包括薄纱敷料)配抗菌剂
- 含铜或银的宫内避孕药.
Operon strategist medical device consultancy provide cost effective and qualitative regulatory service to their clients . we always work hard, do keen research on clients need and give error free deliverable .
Reference:

